2,2,2-trifluoroethyl tosylate trifluoroethylation agent for heteroatom nucleophiles
trifluoroethylation agent for heteroatom nucleophiles; can function as a nucleophile for synthesis of α-keto acids and 2,2-difluorovinyl compounds
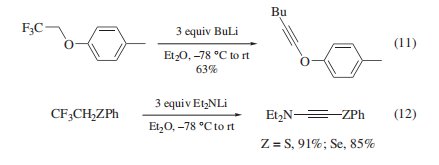
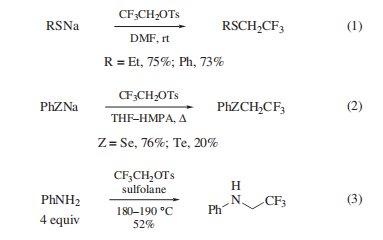
Triflfluoroethylation Agent. Although this reagent is sluggish in substitution reactions compared to its nonflfluorinated counter part,heteroatom nucleophiles react with the reagent to permit introduction of the 2,2,2-triflfluoroethyl group. Thiolsthiophenols, and phenols undergo S- and O-triflfluoroethylation via their sodium salts in good yields (eq 1). For the preparation of alkyl triflfluoroethyl ethers, there is an alternative route via the alkylation of triflfluoroethanol with alkyl halides. ,Triflfluoroethylation of alkali selenolates and tellurolates is also achieved (eq 2), whereas triflfluoroethyl iodide is a better choice in the case of tellurium. N-Triflfluoroethylation is carried out with amines (eq 3) and pyrroles under forcing conditions, while the indole nitrogen is not alkylated but undergoes tosylation instead. In the triflfluoroethylation of less reactive nucleophiles, 2,2,2-triflfluoroethyl tri-flfluoromethanesulfonate generally gives better results, especially in case of nitrogen and carbon nucleophiles.
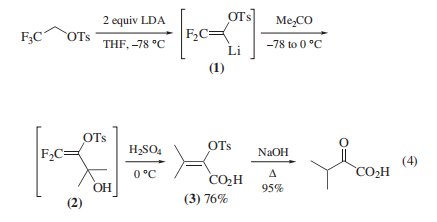
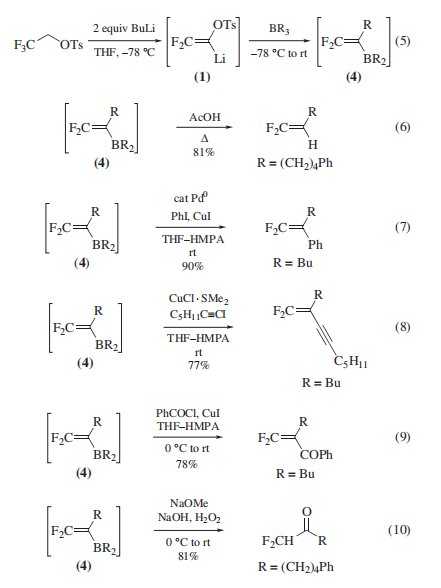
Synthesis of α-Keto Acids and 2,2-Diflfluorovinyl Compounds. The reagent reacts with 2 equiv of Lithium Diisopropylamide10 or Butyllithium1 at −78 ◦C to generate 2,2-diflfluoro- 1-tosyloxyvinyllithium (1) This vinyllithium reagent reacts with carbonyl compounds, to give 3,3-diflfluoro-2-tosyloxyallyl alcohols (2) which undergo Sulfuric Acid-catalyzed hydration to 2-tosyloxyacrylic acids (3) Base-catalyzed hydrolysis of (3) then affords the two-carbon homologated α-keto acids .
Vinyllithium (1) is also utilized via 2,2-diflfluorovinylboranes (4) (eq 5) to synthesize a wide variety of gem-diflfluorovinyl compounds: 1,1-diflfluoro-1-alkenes, 1,1-diflfluoro-1,3-alkadienes, 1,1-diflfluoro-1-alken-3-ynes, 1,1-diflfluoro-2-iodo-1- alkenes, β,β-diflfluoro-α,β-enones, and 2,2-diflfluorovinylphosphines, as well as diflfluoromethyl ketones (eqs 6–10). In these reactions, the reagent functions as a building block for a diflfluorovinylidene unit (CF2=C), to which two different substituents can be successively attached.
In addition, triflfluoroethyl ethers, thioethers, and selenoethers prepared as above are available for the introduction of an alkynemoiety, leading to alkynic ethers and thioethers 3b and thio-3a and selenoynamines6a.
Related Reagents. Chlorodiflfluoromethane; Dibromodiflfluoromethane; 1,2-Diethoxy-1,2-bis(trimethylsilyloxy)ethylene; (Diethoxyphosphoryl)diflfluoromethyllithium; Diethyl Diflfluoromethylphosphonate; Diflfluoromethylenetriphenylphosphorane; Ethyl Diethoxyacetate; Glyoxylic Acid; Glyoxylic Acid Diethyl Dithioacetal; Methyl Bis(2,2,2-triflfluoroethoxy)phosphinylacetate; 2,2,2-Triflfluoroethyl Triflfluoroacetate.