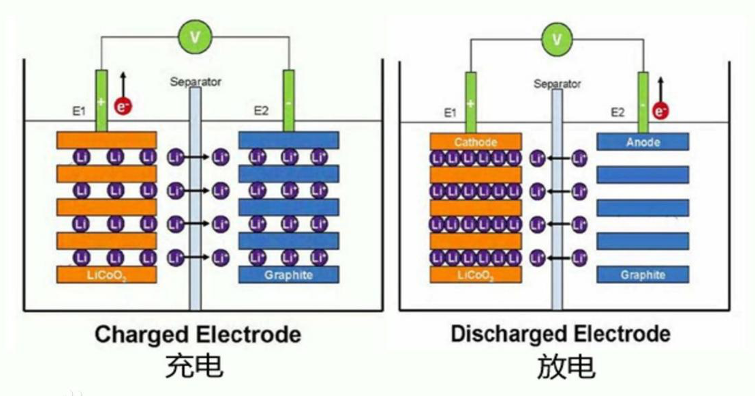
Lithium-ion batteries use lithium alloy metal oxides as the positive electrode material, and mainly use graphite as the negative electrode material. Cathode materials mainly include lithium cobalt oxide (LiCoO2), lithium manganese oxide (LiMn2O4), lithium nickel oxide (LNO), lithium nickel manganese oxide (LiNi0.5Mn1.5O4), lithium iron phosphate (LFP), ternary layered (nickel) Cobalt-manganese NCM or nickel-cobalt-aluminum ternary NCA), lithium-rich manganese (Li-rich or OLO). The negative electrode material is mostly graphite. In addition, lithium metal negative electrode materials, alloy negative electrode materials, tin-based negative electrode materials, oxide negative electrode materials, etc. are also used for the negative electrode. Lithium ions intercalate when the positive electrode reacts and discharge, and lithium ions deintercalate when charging. Lithium ions are deintercalated when the negative electrode reacts and discharges, and lithium ions are intercalated during charging. Lithium-ion batteries can realize secondary charge and discharge through the transfer of lithium ions between the positive and negative electrodes. Take the cathode material lithium cobalt oxide as an example, when charging: LiCoO4 → Li1-xCoO4 + xLi+ + xe-; when discharging: Li1-xCoO4 + xLi+ + xe- → LiCoO4. When charging: xLi+ + xe- + 6C → LixC6; when discharging: LixC6→xLi+ + xe- + 6C. As shown below:
2. Non-aqueous liquid electrolyte
The electrolyte plays the role of conducting electrons between the positive and negative electrodes of the lithium battery, which is the guarantee for the lithium ion battery to obtain the advantages of high voltage and high specific energy. Liquid electrolytes are generally prepared from raw materials such as high-purity organic solvents, electrolyte lithium salts, and necessary additives under certain conditions and in certain proportions.
The liquid electrolyte materials used should generally have the following characteristics: ① High conductivity, low electrolyte viscosity, high lithium salt solubility and ionization degree; ② High Li+ conductive migration number; ③ High stability, requiring electrolyte with high flash point, High decomposition temperature, low electrode reaction activity, no side reactions, long time, etc.; ④The interface is stable, and it has good film-forming characteristics on the surface of positive and negative materials. It can form a stable during the first few cycles of charge and discharge. Low-impedance solid electrolyte interphase (solid electrolyte interphase, SEI film); ⑤Wide electrochemical window, can passivate the electrode surface, so as to work in a wide voltage range; ⑥Wide operating temperature range; ⑦and positive and negative electrodes The material has good wettability; ⑧ not easy to burn; ⑨ environmentally friendly, non-toxic or low toxicity; ⑩ lower cost.
1. Solvent
The solvent plays a role in dissolving the electrolyte lithium salt and transferring lithium ions. The properties of lithium ion battery electrolytes are closely related to the properties of the solvent. Generally speaking, the choice of solvent should meet the following basic requirements: ① An organic solvent should have a high dielectric constant, so that it can dissolve the lithium salt high enough ②The organic solvent should have a lower viscosity, so that Li+ in the electrolyte is easier to migrate; ③The organic solvent must be inert to each component in the battery, especially in the battery working voltage range must be with the positive electrode and The negative electrode has good compatibility; ④The organic solvent or its mixture must have a lower melting point and a higher boiling point, in other words, a relatively wide liquid range, so that the battery has a relatively wide operating temperature range; ⑤The organic solvent must have a relatively high temperature The safety (high flash point), non-toxic and harmless, low cost.
Since the solvent needs to have a higher dielectric constant, the organic solvent that can be used in lithium-ion batteries should contain polar groups such as carboxyl groups, nitrile groups, sulfonyl groups, and ether chains. The research on lithium-ion battery solvents mainly focuses on organic ethers and organic esters, which are divided into cyclic and chain forms.
Cyclic ethers mainly include tetrahydrofuran, 1,3-dioxolane and 4-methyl-1,3-dioxolane. Cyclic ethers are generally mixed with organic ester solvents such as dimethyl carbonate and propylene carbonate, because they are prone to ring-opening polymerization in the electrolyte and do not have stable electrochemical performance, which largely limits their use in the electrolyte. In the application. Chain ethers mainly include dimethoxymethane (DMM), 1,2-dimethoxyethane (DME), 1,2-methoxypropane (DMP), etc. Generally speaking, the longer the carbon chain of the electrolyte solvent, the stronger its oxidation resistance, but at the same time the higher the viscosity and the lower the conductivity. Chain ethers can react with the cations of lithium hexafluorophosphate (LiPF6) in the electrolyte to form a stable chelate, which increases the solubility of the lithium salt, thereby increasing the conductivity of the electrolyte, but it has strong reactivity with lithium, making it difficult A stable SEI film is formed. Therefore, ether solvents are mainly used in disposable lithium batteries.
Ester solvents mainly include two types of carboxylic acid monoesters and carbonic acid dibasic esters. The most important cyclic carboxylic acid ester is γ-butyrolactone (BL). Generally speaking, as the number of carbon atoms increases, the dielectric constant, melting point, and viscosity of the cyclic carboxylic acid ester will decrease, and BL is easily decomposed in water, has high toxicity, and the cycle efficiency is much lower than that of carbonate organic solvents, so Such macromolecular cyclic carboxylic acid esters are mostly used in disposable batteries. The common chain carboxylic acid esters are methyl formate, ethyl formate, ethyl acetate and so on. Chain carboxylic acid ester has the advantages of lower melting point, easy purification, higher dielectric constant, etc., but due to its strong polarity, it is easier to react with lithium, which is not conducive to the cycle performance of the battery. Carbonate is currently the most common and mature commercial electrolyte solvent on the market. The most commonly used cyclic carbonates are ethylene carbonate (EC) and propylene carbonate (PC). EC has higher thermal stability, dielectric constant and lower viscosity, which is beneficial to the dissolution of lithium salt and ion conduction, and is beneficial to the formation of SEI film. However, the higher melting point of EC affects the low-temperature performance of the battery. PC has a high dielectric constant and chemical and electrochemical stability, but before the formation of the negative SEI film, PC is likely to be embedded in the graphite layer with lithium, causing the graphite layer to peel off, resulting in a decrease in the cycle performance of the battery. Chain carbonates mainly include dimethyl carbonate (DMC), diethyl carbonate (DEC), and ethyl methyl carbonate (EMC), which have lower viscosity and lower dielectric constant. DEC and DMC alone cannot form a stable SEI film on the surface of lithium, while EMC has poor thermal stability.
It is difficult for a single solvent to meet the various performance requirements of the electrolyte. Therefore, in practical applications, several organic solvents with different properties are usually mixed and used. For example, for organic esters, most of the cyclic organic esters have a wider liquid range, higher dielectric constant, and higher viscosity, while chain solvents have a narrower liquid range and lower medium. Electric constant and low viscosity, so generally chain and cyclic organic ester mixtures are used in the electrolyte as the solvent of the lithium-ion battery electrolyte.
2. Lithium salt
Lithium salt is the main provider of lithium ions in the electrolyte. The lithium salt used in lithium-ion batteries needs to meet the following basic requirements: ①It has relatively high solubility in organic solvents and is easy to dissociate, thereby ensuring that the electrolyte has a relatively high ②It has relatively high oxidation-reduction stability, and does not undergo electrochemical and thermodynamic reactions with organic solvents, electrode materials and battery components; ⑧Lithium salt anions are non-toxic, harmless and environmentally friendly; ④The production cost is low, Easy to prepare and purify.
Lithium salts frequently studied are mainly compounds based on mild Lewis acids. These compounds mainly include lithium phosphate (LiPF6), lithium borate (LiBF4), lithium perchlorate (LiClO4), lithium arsenate (LiAsF6), and lithium antimonate. (LiSbF6), etc.; and some organic lithium salts, such as LiCF3SO3, LiN(SO2CF3)2 and their derivatives. Lithium hexafluorophosphate (LiPF6) is currently a widely used electrolyte lithium salt in commercial lithium-ion batteries. Its comprehensive performance has the most advantages. The main drawback is that it is unstable at high temperatures and sensitive to water. Lithium tetrafluoroborate (LiBF4) has good overall performance, among which the high and low temperature performance is better than LiPF6, but its conductivity is not high and it is easy to react with lithium metal. Many lithium salts generally have relatively good overall performance, but they all have some serious shortcomings, which limit their large-scale application.
3. Electrolyte additives
Commercial lithium-ion batteries may contain more than 10 kinds of additives, these additives are characterized by a small amount but can significantly improve the performance of a certain aspect of the electrolyte. Their functions generally include improving the conductivity of the electrolyte, improving the cycle efficiency of the battery, increasing the reversible capacity of the battery, improving the film-forming performance of the electrode, and improving the safety of the battery.
1) Film-forming additives
Generally, the lithium intercalation potential of negative electrode materials (such as carbon, silicon, etc.) is lower than 1.2 V vs. Li+/Li. During the first charging of lithium-ion batteries, it will inevitably react with the electrolyte. Solvent molecules, lithium salts or additives It is reduced at a certain potential and produces some insolubles, thus forming a layer of SEI film on the surface of the negative electrode. The specific components of the SEI film are very complex and vary depending on conditions such as battery materials and temperature. It is a layer of electronically insulating and ion-conducting membrane, which can prevent further reaction between the electrolyte and the negative electrode material without affecting the passage of Li+. The formation of SEI film has a non-negligible effect on improving the properties of the electrode and increasing the life of the electrode. However, the presence of the SEI film will reduce the electronic contact between the electrode material particles and reduce the efficiency of the first week. Therefore, it is generally undesirable for the thickness of the SEI film to be large.
There may also be an interface film on the surface of the positive material, which has similar properties to the negative SEI film and can prevent further reactions between the electrolyte and the electrode material, but its formation mechanism is still controversial.
The film-forming additive is easier to be reduced on the negative electrode than the solvent. Therefore, after being added to the electrolyte, it will be reduced on the negative electrode in preference to solvents such as EC, participating in the formation of the SEI film, and the resulting film has good ion permeability. Good electronic insulation is good for lithium ions to enter and exit the negative electrode during charge and discharge, but the electrons on the negative electrode cannot contact solvent molecules, which improves the rate performance and also improves storage, low-temperature discharge and high-temperature charge and discharge performance.
Film-forming additives are mainly divided into inorganic film-forming additives (such as SO2, CO2, CO and other small molecules and lithium halides, etc.) and organic film-forming additives, such as vinylene carbonate (VC), propylene sulfite (PS), and sulfurous acid Vinyl esters (ES), etc., also include some halogenated carbonates.
2) Ion conductive additives
The role of ion conductive additives is to improve the conductivity of the electrolyte. The research on improving the conductivity of the electrolyte mainly focuses on promoting the dissolution and ionization of conductive lithium salts and preventing the damage of the electrode by the co-intercalation of the solvent.
According to the type of action, it can be divided into cation action type (mainly including some amines, aromatic heterocyclic compounds with more than two nitrogen atoms in the molecule, crown ether and cryptate), and anion action type (mainly some anions are affected by Body compounds, such as boron-based compounds), and the type that interacts with electrolyte ions (neutral ligand compounds are mainly compounds formed by electron-rich groups bonded to electron-deficient atoms N or B, such as azaethers and alkyl borons ). Currently commonly used conductive additives include 12-crown-4 ether, anion acceptor compounds and inorganic nano-oxides.
3) Flame retardant additives
Safety has always been the most concerned issue of lithium-ion batteries, and the addition of flame retardant additives can improve the safety of the electrolyte to a certain extent. Its main mechanism is that the electrolyte of a lithium ion battery will undergo a chain accelerated reaction initiated by free radicals when heated, and the flame retardant additive can capture free radicals and block the chain reaction.
The main types are organophosphorus compounds, organofluorinated compounds, halogenated alkyl phosphates and ionic liquids. Commonly used flame retardant additives include phosphate esters such as trimethyl phosphate (TMP) and triethyl phosphate (TEP), fluorocarbon esters such as methyl difluoroacetate (MFA) and ethyl difluoroacetate (EFA) and ionic liquids. Wait.
4) Overcharge protection additive
When a lithium-ion battery is overcharged, a series of reactions occur. The first is that too much Li+ is extracted from the positive electrode material and embedded in the negative electrode material, which may lead to the collapse of the positive electrode material structure and the formation of negative electrode lithium dendrites. This mainly occurs in the positive electrode material where Li cannot be completely extracted, such as LiCoO2, Li1+ x(NiCoMn)O2, etc.; followed by the irreversible oxidation and decomposition reaction of electrolyte components (mainly solvent) on the surface of the positive electrode, generating gas and releasing a large amount of heat, which leads to an increase in the internal pressure and temperature of the battery, which contributes to the safety of the battery. Sex has a serious impact.
The overcharge problem can be solved by the control and protection of the external circuit. When the charging voltage reaches 4.1~4.2V, the control voltage of the electronic integrated circuit will no longer rise. In addition, the use of overcharge protection additives is also an effective method. Its basic principle is: the oxidation potential of the additive is slightly higher than the positive electrode delithiation potential. When the battery exceeds the working voltage, the additive reacts first, causing the battery to open or short circuit, so that the battery stops working and slowly releases heat. This process Does not damage the electrode material and electrolyte. In the normal working voltage range, the additives do not participate in the battery reaction. Overcharge protection additives mainly include three types of additives: redox couple, electropolymerization and gas generation. Among them, the redox couple additive is the most commonly used. The principle is: when the voltage exceeds the battery cut-off voltage, the additive is oxidized on the positive electrode surface, the oxidation product diffuses to the negative electrode surface to be reduced, and the reduction product diffuses to the positive electrode surface to continue to be oxidized. The process cyclically continues until the end of charging. The best overcharge protection additive should have a cut-off voltage of 4.2~4.3V, so as to meet the requirement of a lithium-ion battery with a voltage greater than 4V.
The more common overcharge protection additives include ortho and para dimethoxy substituted benzene, butyl ferrocene and biphenyl.
5) additives to control the acid and water content in the electrolyte
Because lithium and water will react violently immediately upon contact, all materials and parts including electrolytes must be strictly dehydrated and reliably sealed. However, even a small amount of HF and moisture in the electrolyte will cause the decomposition of LiPF6 and the destruction of the electrode material surface, so further control of the moisture and acid content in the electrolyte is necessary. The mechanism of action of these additives is mainly to reduce their content by combining with the acid and water in the electrolyte.
At present, the commonly used additives to control the content of HF mainly include lithium or calcium carbonate, aluminum oxide, magnesium oxide and barium oxide, which can react with a small amount of HF in the electrolyte to prevent its influence. However, these substances are slow to remove HF, so it is difficult to prevent HF from damaging the battery performance. Although some acid anhydride compounds can quickly remove HF, they will also produce other acidic substances that damage battery performance. The additives that control the water content are mainly hexamethyldisilane (HMDS), alkanediimine and other highly water-absorbing compounds.
6) additives to improve low temperature performance
Low-temperature performance is one of the important factors to expand the use range of lithium-ion batteries. At low temperatures, the electrolyte has problems such as increased viscosity, decreased conductivity, and increased impedance of the SEI film, which greatly weakens the energy density and cycle performance of the battery. At present, the conventional electrolyte components are LiPF6 as the electrolyte salt, ethylene carbonate (EC) as the solvent matrix, and mixed solvents (such as DMC, DEC, EMC, etc.) are added. This type of electrolyte has the advantages of high discharge capacity, high conductivity, low internal resistance, fast charge and discharge rate, and good compatibility with electrode materials, but EC has a relatively high freezing point (36°C), so low temperature performance is poor. At present, the main ways to improve the low-temperature performance of the electrolyte are as follows: One is to reduce the content of EC by adding low-melting, low-viscosity components to reduce the viscosity and eutectic point of the electrolyte at low temperatures. The second is to use electrolyte lithium salt that can maintain a small charge transfer resistance at low temperatures. The third is to add some film-forming additives to optimize the composition of the SEI film to reduce its resistance at low temperatures.
Commonly used additives to improve high and low temperature performance include N,N-dimethyl trifluoroacetamide, methyl ethylene carbonate (MEC) and fluoroethylene carbonate (FEC).
7) High voltage additives
The use of high-voltage additives is an effective means to improve the oxidation resistance of carbonate system electrolytes. This type of additive is mainly a positive electrode film-forming additive. Its mechanism of action is similar to that of a negative electrode film-forming additive. Its HOMO energy is slightly higher than that of solvent molecules. Form a film on the surface of the positive electrode in advance to improve the surface properties of the positive electrode material and reduce the contact between the positive electrode material and the electrolyte to achieve the purpose of inhibiting the decomposition of the electrolyte.
At present, the commonly used high-voltage additives mainly include benzene derivatives (such as biphenyl, terphenyl), heterocyclic compounds (such as furan, thiophene and their derivatives), 1,4-dioxane and hexafluorotriphosphate Isopropyl ester and so on. They can effectively improve the oxidation stability of electrolyte under high voltage, and play a very important role in high voltage lithium ion batteries.
In addition, studies have found that the purity of the solvent also has an important effect on the oxidation resistance of the electrolyte, and the improvement of the solvent purity can greatly improve the oxidation resistance of the electrolyte. For example, when the purity of EC increases from 99.91% to 99.979%, its oxidation potential increases from 4.87 V to 5.5 V vs. Li+/Li.
There are many types and functions of additives. In addition to those described in detail above, there are many other functional additives, such as LiBOB and LiODFB and other additives to inhibit corrosion of aluminum foil, and additives such as biphenyl and o-terphenyl to improve the film-forming performance of positive electrodes. , Tris (2,2,2-trifluoroethyl) phosphite (TTFP) and other additives to improve the stability of LiPF6. In fact, some additives themselves are multifunctional additives. For example, after 12-crown-4 ether is added to the PC solvent, it can improve the conductivity of Li+ and use the electrophilic effect of the crown ligand on the electrode surface to make Li+ on the electrode surface. The possibility of the interface reacting with solvent molecules is greatly reduced. The desolvation effect of crown ether on Li+ inhibits the co-intercalation of PC molecules. The SEI film at the electrode interface is optimized to reduce the first irreversible capacity loss of the electrode; fluorinated organic solvents, halogenated After the phosphate is added to the electrolyte, it not only helps to form an excellent SEI film, but also has a certain or even obvious flame-retardant effect on the electrolyte, which can improve the performance of the battery in many aspects.
3. Ionic liquid electrolyte
Ionic liquid electrolyte, that is, molten salt at room temperature, is composed entirely of anions and cations. Due to the large volume of anions or cations, the interaction force between the anions and cations is weak, and the electron distribution is uneven. The anions and cations can move freely at room temperature and are in a liquid state. Compared with lithium-ion batteries, ordinary organic solvent ionic liquids have the following advantages: ①The vapor pressure of ionic liquids is very low, almost negligible; ②Ionic liquids have a wide range of about 300℃; ③Ionic liquids are not flammable; ④ It has high conductivity; ⑤Good chemical or electrochemical stability; ⑥Insensitive to water and air; ⑦No pollution and easy to recycle.
There are many types of ionic liquids, which can be divided into the following categories according to the different organic cations: nitrogen-containing heterocycles, quaternary ammonium salts and quaternary phosphorus salts, etc., among which nitrogen-containing heterocycles include imidazole salts and piperidines Salts and pyridine salts, etc. There are also many types of anions that make up ionic liquids. According to the random combination of anions and cations, in theory, thousands of ionic liquids can be composed.
However, ionic liquid electrolytes also have many problems. The first is that most ionic liquids have high viscosity, which is 1 to 2 orders of magnitude higher than that of ordinary organic solvents, resulting in poor rate performance of the battery; secondly, the compatibility with the positive and negative materials is poor. It is difficult to use liquid electrolytes; in addition, ionic liquids are currently expensive, which limits their large-scale application in lithium-ion batteries.
Reduce the viscosity of the ionic liquid and increase the migration rate of lithium ions in it. It is the ionic liquid electrolyte that can
The prerequisite for commercialization in lithium-ion batteries. At present, there are two main ways to solve this problem: One is to blend ionic liquids with organic solvents or other materials to form a solvent system. The three types of electrolytes formed by mixing ionic liquids with carbonate electrolyte solutions, polymers or inorganic ceramics have received a lot of research and achieved good results. The development of these new systems is expected to promote the practical application of ionic liquids in batteries. The second is to adjust the anion and cation structure of the ionic liquid. Studies have found that the use of ionic liquids with asymmetric structures can help reduce their viscosity.
Four, gel polymer electrolyte
Gel-type polymer electrolyte is usually defined as: polymer as the backbone of the entire electrolyte plays a role of mechanical support, and its network structure enables the electrolyte formed by alkali metal salts and organic plasticizers to be uniformly distributed in the network , And the transport of ions mainly occurs in the part containing the liquid electrolyte. The conductivity of this type of electrolyte is equivalent to that of an organic solution of an alkali metal salt, and can generally reach more than 10-3 S/cm at room temperature. Commonly used gel polymer electrolyte matrixes are: polyacrylonitrile (PAN), polyethylene oxide (PEO), polymethyl methacrylate (PMMA), polyvinylidene fluoride (PVDF), etc. The conductivity of gel polymer electrolytes is also related to the types of organic solvents. Organic solvents such as EC, PC, and BL are commonly used as plasticizers.
Five, high voltage electrolyte
Further improving the energy density of lithium-ion batteries has always been the goal of lithium-ion battery research. Under the existing lithium-ion battery system, there are two main ways to increase energy density: ①Use high-voltage cathode materials, such as spinel LiNi0.5Mn1.5O4 and olivine structure LiCoPO4; ②Use high-capacity cathode materials, Such as lithium-rich phase positive electrode. Both of these electrode materials have a relatively high lithium removal potential, and ordinary carbonate electrolytes are easily oxidized and decomposed. The development of high-voltage electrolytes with a wider electrochemical window is an important direction for the development of electrolytes for lithium-ion batteries. At present, high-voltage electrolytes mainly include the following systems.
1. Sulfone based electrolyte
The sulfonyl group of sulfones has stronger electron-withdrawing ability than the carbonyl group of carbonate, so the energy level of the occupied molecular orbital (HOMO) is lower. The oxidation potential of most sulfone solvents can reach 5.5 V (vs. Li+/ Li) above, it has excellent electrochemical stability; and the sulfone solvent itself has strong flame retardancy. Therefore, sulfone solvents have also been extensively studied in lithium batteries. Based on ethyl methyl sulfone and tetramethyl sulfone (TMS), which have relatively high ionic conductivity and relatively wide electrochemical window, they have good compatibility with high voltage cathode materials such as LiMn2O4 and LiNi0.5Mn1.5O4 . However, the melting point of ethyl methyl sulfone (EMS) is relatively high (36.5°C), and the low temperature performance is not very good. Methoxyethyl methyl sulfone (MEMS) has a relatively low melting point (about 0°C). MEMS-based electrolytes use LiTFSI as the lithium salt, which has a relatively wide electrochemical window, but has poor compatibility with graphite anodes .
Many problems of sulfone electrolytes limit its application: the current synthesis of sulfone solvents is relatively complicated; most of the melting points are higher than room temperature and the viscosity is large; the compatibility with graphite anodes is relatively poor, and it cannot form stable on the surface of graphite anodes. SEI film; and the poor wettability with Celgard separator, it is necessary to use a more expensive glass fiber separator to assemble the battery. Adding film-forming additives or sharing with carbonate solvents can improve some of the problems.
2. Fluorine-substituted carbonate system electrolyte
Fluorine-substituted carbonate has the advantages of low melting point, high oxidation stability and non-flammability, and its cycle performance and storage performance are significantly improved at high temperatures. Many researchers use it as an electrolyte additive or co-solvent for lithium-ion batteries. Electrolyte composed of fluoroethylene carbonate (FEC), difluoroethyl acetate (DFEAc), bis(2,2,2-trifluoroethyl) carbonate (DTFEC) and other fluorocarbonate and conventional carbonate solvents , Usually has the advantages of high conductivity, high oxidation stability, good electrode compatibility, etc., showing excellent cycle performance and safety performance. Fluorine-substituted carbonate is expected to be used in electrolyte solvents for high-voltage systems, but its drawback is that it has poor solubility for LiPF6 and needs to be further improved.
3. Nitrile electrolyte
Nitrile solvents have higher lightning, wider liquid range, higher dielectric constant and lower viscosity. The terminal nucleophilic nitrile group of nitrile substances is the main site of lithium ion coordination, which makes the solubility of lithium salt in nitrile solvents higher, so nitrile electrolytes usually have higher ionic conductivity. The oxidation potential of glutaronitrile and adiponitrile is as high as 8.3 V vs Li+/Li, which is known as the solvent with the highest oxidation potential. Nitriles such as glutaronitrile, adiponitrile and sebaconitrile as a co-solvent and carbonate solvent composition of the electrolyte has higher conductivity, lower viscosity and stronger oxidation resistance, and LiNi0.5Mn1. High voltage cathode materials such as 5O4 and Li2NiPO4F have good compatibility. In addition, the nitrile electrolyte also has excellent low temperature performance. However, the compatibility of nitrile solvents with the negative electrode is poor, and it is necessary to use carbonate as a co-solvent or use film-forming additives such as VC and LiBOB to improve its performance.
6. Introduction of Fluorinated Solvents or additives
Fluorinated solvents or additives are important materials for the development of lithium-ion battery electrolytes with high performance and special functions. There are 7 electrons in the outermost layer of the electron orbit of fluorine, which has strong electronegativity and weak polarity. Fluorine will reduce the freezing point of solvents, increase lightning, increase oxidation resistance, and increase flame retardancy, which helps to improve The contact performance between the electrolyte and the electrode, and is beneficial to the formation of a better protective film on the positive and negative electrodes. Therefore, the use of fluorinated solvents or additives in the electrolyte will effectively improve the low-temperature performance, oxidation resistance, flame-retardant performance, wettability and cycle performance of the counter electrode, and then obtain electrolytes with special functions, such as high Voltage electrolyte, flame retardant electrolyte, wide temperature window electrolyte, etc.
When used as an additive, the amount of each additive generally does not exceed 10 wt.%, and in most cases it does not exceed 5 wt.%.
For specific research and application examples, see the table "Research and Application of Several Fluorinated Electrolyte additives".
Contact: Mr Hsu
Phone: +86(0)1815701-8567
Tel: +86(0)570780-5618
Email: sales@fluorio.com
Add: No.5,Longfei Road,Longyou Eco-Tech Development Zone, Quzhou, Zhejiang, China