Highly regioselective electrochemical fluorination of 2-pyrimidyl sulfides having an electron-withdrawing group (EWG) at the position α to the sulfur atom was successfully carried out using Et4NF·nHF (n = 3, 4) or Et3N·3HF as a supporting electrolyte and a fluoride ion source in 1,2- dimethoxyethane (DME) in an undivided cell. 2-Methylthiopyrimidine devoid of an EWG was also selectively fluorinated in DME to provide 2- (fluoromethylthio)pyrimidine in a moderate yield as 63%, while corresponding 2-methylthiopyridine was less selectively fluorinated in lower yield along with α,α-difluorinated product. In contrast, the corresponding 2- quinazolinonyl sulfides underwent similarly α-fluorination along with unexpected ipso-fluorination through anodic desulfurization.
Yi Cao,Katsutoshi Suzuki,Toshiki Tajima...
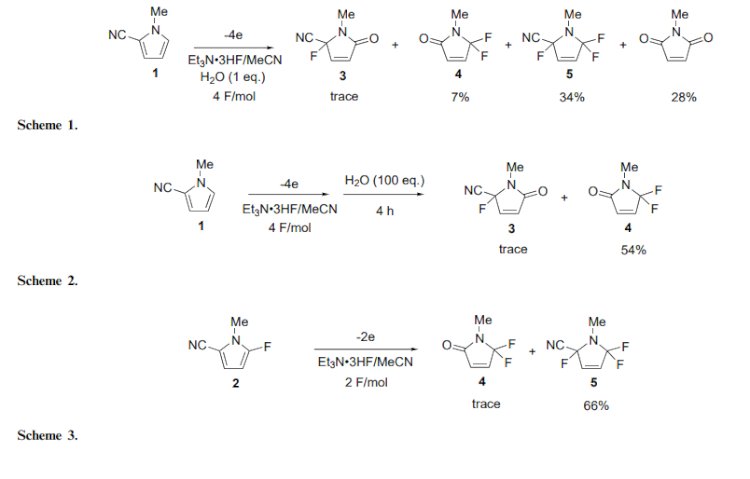
Fluorinated aromatic heterocyclic compounds have been of much interest because of their unique biological properties.2 Although direct fluorination of heteroaromatic rings is the simplest way to prepare fluorinated heterocycles, successful examples of the fluorination are limited. For example, the chemical fluorination of fivemembered heteroaromatic compounds such as pyrroles, thiophenes and furans were attempted; however, the yields were extremely low (less than 6%) and the selectivity was also unsatisfactory in all cases.3 Recently, electrochemical partial fluorination of organic compounds has been shown to be a new powerful method for selective fluorination.4 However, there have been few reports of direct fluorination of heteroaromatic compounds using an electrochemical method.5 In most cases, yields of the fluorinated product are extremely low or unsatisfactory. On the other hand, we found that electron-withdrawing groups promoted the anodic fluorination of heterocyclic compounds.5,6 With these facts in mind, we attempted the direct anodic fluorination of pyrroles having an electron-withdrawing cyano group.

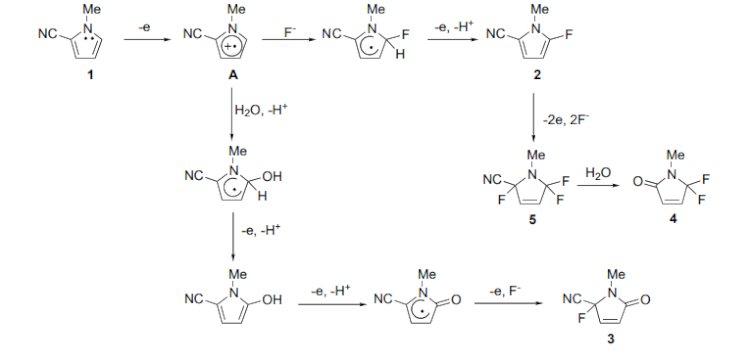
In conclusion, we have successfully carried out for the first time anodic fluorination of 2-cyano-1- methylpyrrole. The product 4 has a biologically interesting gem-difluoromethylene unit in the heterocyclic ring and 2 has also a versatile cyano group and a fluorine atom. Therefore, 2 and 4 seem to be useful fluorinated building blocks. Work on further application and the scope and limitation of the methodology for anodic fluorination is now in progress.
Contact: Mr Hsu
Phone: +86(0)1815701-8567
Tel: +86(0)570780-5618
Email: sales@fluorio.com
Add: No.5,Longfei Road,Longyou Eco-Tech Development Zone, Quzhou, Zhejiang, China